Don't say it don't use it - Alzheimer's treatments and the stats vs effect dilemma
Katrina Borthwick - 28th April 2026
The Cochrane Reports are comprehensive reviews that summarise and evaluate the results of clinical research on healthcare interventions. They are produced by an international network of researchers and healthcare professionals called the Cochrane Collaboration. These reports are widely respected for their sound scientific approach, and often include meta-analyses of multiple studies.
A recent Cochrane Report published on 16 April 2026 dug into the clinical benefits and harms of amyloid‐beta‐targeting monoclonal antibodies (Aβ-mAbs) in people with mild cognitive impairment or mild dementia due to Alzheimer’s disease.
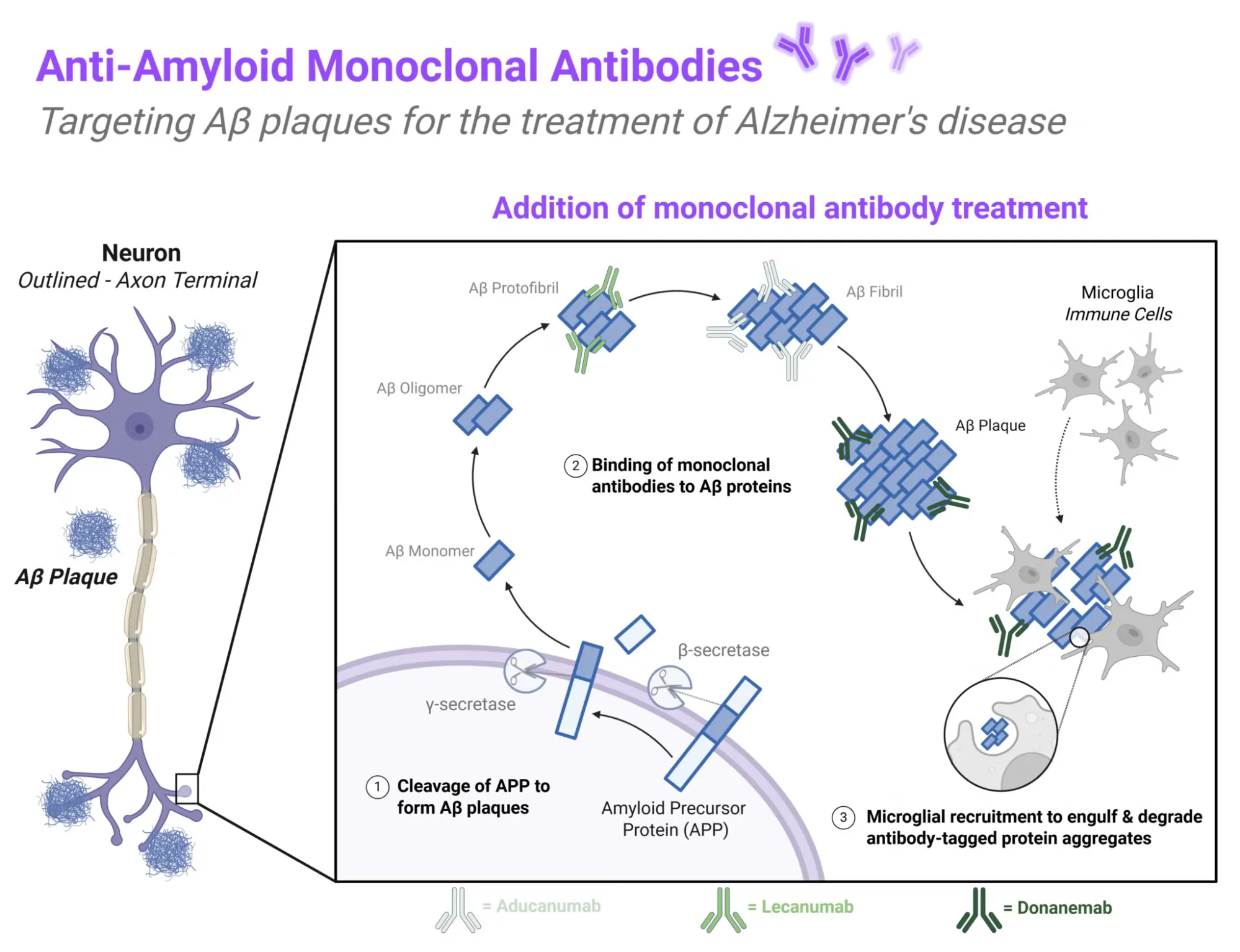
The theory was that these plaques were causing Alzheimer’s symptoms. So reducing the plaques by introducing antibodies that engulf and degrade them could be a viable Alzheimer’s treatment. There is a heap of research on this, and the Cochrane report had a look at a wide range of these studies to see if the results held up.
Clinical effect
Clinical assessments of Alzheimer treatments focus on three measures - cognition, dementia and functional ability.
In terms of cognition, the “Alzheimer’s Disease Assessment Scale - Cognitive” (ADAS-Cog) scale provides the most recognised measure, and tests memory, language, praxis (including tasks like copying drawings or addressing envelopes) and awareness of time and space. It asks participants to complete tasks like remembering words and following instructions. According to 13 studies involving 9,895 people using the ADAS-Cog, Aβ-mAbs probably makes little to no difference in cognitive abilities compared to a placebo.
For dementia, the “Clinical Dementia Rating - Sum of Boxes” (CDR-SB) scale is used to measure the severity of dementia. It works by scoring six different areas of a person’s abilities, including memory, orientation, judgment, community affairs, home and hobbies, and personal care. Each area is rated, and the scores are added together to give an overall picture of how much dementia is affecting someone’s daily life and independence. The Cochrane review found that across nine studies involving 8,053 people using the CDR-SB, Aβ-mAbs probably makes little or no difference in how severe dementia becomes - although this conclusion is less certain than the others.
For functional ability, three scales are often used - the “Alzheimer’s Disease Cooperative Study-Activities of Daily Living” (ADCS-ADL) scale and two variants focused on “Instrumental Activities of Daily Living” (ADCS-iADL) and an inventory of daily tasks for “Mild Cognitive Impairment” (ADS-ADL-MCI). The ADCS-ADL scale is a questionnaire used to assess how well people with Alzheimer’s can perform everyday tasks, such as eating, dressing, bathing, and managing household chores. The ADCS-iADL scale focuses on the performance of more complex daily tasks, such as managing money, using the phone, shopping, or preparing meals. The third scale has questions tailored to more mild impairment.
The Cochrane review concluded that Aβ-mAbs probably make little or no difference in how well people with mild Alzheimer’s can do everyday tasks featured in the ADS-CL. There might be a slight improvement in more complex daily activities, as shown by the ADCS-iADL scale and the ADCS-ADL-MCI scale, but the evidence for this is not very strong.
Side effects
So, is it worth giving a go? Well, for treatments with a borderline or questionable positive effect, the main thing we need to be sure of is that they do not cause any harm (including to your wallet).
It appears from the research that Aβ-mAbs are responsible for a small increase in the chance of developing brain swelling, with this occurring in about 107 more cases per 1,000 people than placebo, based on moderate certainty from 11 studies involving 13,595 participants. However, most of these observations were non-symptomatic, with only about 29 more cases per 1,000 than placebo having symptomatic brain swelling or brain bleeds.
In terms of side effects, the study found that after 18 months, the treatments show a very small increase in the risk of serious side effects (about 6 more events per 1,000 people) and overall death rates (about 2 more deaths per 1,000 people). This is not much, but if the benefits are not proven, it would seem to be an unnecessary risk.

Statistical significance versus clinical significance
Statistical significance and clinical significance are both important concepts in medical research, but they refer to different things.
Statistical significance means that the observed effects in a study are unlikely to have occurred by chance alone. However, statistical significance does not necessarily mean that the clinical effect is big enough to be meaningful.
Clinical significance refers to treatment outcomes for patients. An outcome can be statistically significant but still so small that it does not make a noticeable difference in a patient’s symptoms. For example, a drug might slightly improve test scores or slow disease progression in a way that is statistically detectable, but the magnitude of this change may not be enough to improve the person’s life meaningfully, or to justify potential side effects and costs.
This distinction is crucial in interpreting medical research, especially in the context of treatments for conditions like Alzheimer’s disease. A treatment may produce statistically significant results in tests, but if these improvements are too minor to alter a person’s ability to function independently or enjoy life, they may not be considered clinically significant. Therefore, it is important to consider whether a result translates into real, meaningful benefits for patients.
The Cochrane Review’s conclusions
The effect of Aβ-mAbs on cognitive function and dementia severity at 18 months in people with mild cognitive impairment or mild dementia due to Alzheimer’s disease is negligible at best. Aβ-mAbs increase the risk of brain swelling and bleeds.
Even though these drugs can remove amyloid plaques from the brain, this does not lead to meaningful improvements for people with mild memory loss or mild Alzheimer’s disease. They suggest future research should look for treatment options that focus on other tactics than eliminating the plaques, suggesting that they don’t think these plaques are the cause of the Alzheimer’s symptoms - as removing them has no clinical effect.
Critics and comments
The reaction to this paper has been mixed. John Hardy, the British biologist who first developed the amyloid hypothesis in the 90s, is a bit peeved, and criticised the review for lumping together data about lecanemab and donanemab, along with drugs that are known to be ineffective, therefore dragging down the overall average. He told Agence France Press (AFP) “This is a silly paper which should not have been published”.
The authors responded that while the drugs included in the study may work in different ways, they all target amyloid-beta proteins.
Australian neuroscientist Bryce Vissel, who was not involved in the research but has written papers about anti-amyloid antibody treatments, chimed in and said it “does not prove amyloid has no role in Alzheimer’s, and it does not rule out future amyloid-directed therapies that may yet help patients… But it does show that the current generation of anti-amyloid drugs is not delivering the promise that has surrounded it.”

So should we be using statistical significance at all?
According to an article in the Australian Prescriber, perhaps we should stop using statistical significance in clinically relevant studies. It turns out it is not just laypeople taking statistical significance as proof a treatment works. Clinicians and the authors of medical literature are also making the same error, and equating statistical significance with proof that a treatment works. They argue that a statement such as “both regimens were [statistically] significantly better than the placebo group” is exactly the same as saying “both regimens were mathematically unusually better than the placebo group”, which is meaningless in the treatment context.
They quote Ron Wasserstein, the Executive Director of the American Statistical Association, as saying “statistically significant - don’t say it and don’t use it”. I figure if anyone has skin in the game on the use of statistical significance, it’s him!
